The TIGRIS trial results have been published in The Lancet Respiratory Medicine
The TIGRIS trial represents a milestone for adjunctive therapies in septic shock, demonstrating the efficacy of Polymyxin B Hemoadsorption (PMX-HA; Toraymyxin, Toray Medical Inc, Tokyo) in improving the outcome in patients with Endotoxic Septic Shock (ESS), selected using Endotoxin Activity Assay (EAA; Spectral Medical Inc., Toronto).
The Tigris trial was a US-based, multicenter, randomised, controlled Phase 3 study evaluating PMX-HA in adults with endotoxic septic shock, defined by an EA level between 0.60 and 0.89 and a Multiple Organ Dysfunction Score (MODS) > 9 or a Sequential Organ Failure Assessment (SOFA) score > 11.
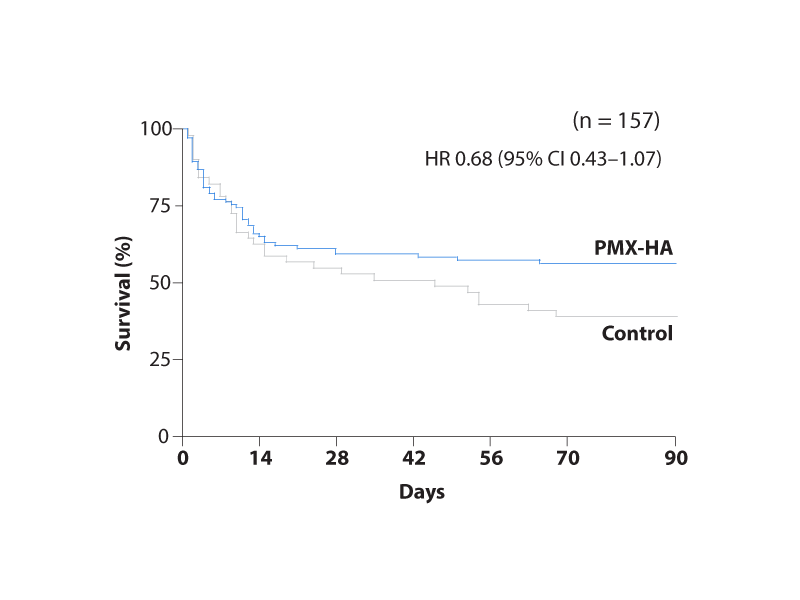
A total of 157 patients were randomised in a 2:1 ratio to receive either PMX-HA plus standard-of-care (n=106) or standard-of-care alone (n=51).
The PMX-HA treatment group achieved the primary and key secondary endpoints, showing:
• 28-day mortality: absolute risk reduction of 6.4% (posterior probability of benefit 95.3%)
• 90-day mortality: absolute risk reduction of 17.4% (posterior probability of benefit 99.4%)
In conclusion, in patients with endotoxic septic shock, Polymyxin B Hemoadsorption is associated with a high probability of lower mortality at 28 and 90 days. The Tigris trial shows a clinically meaningful signal of reduced mortality with polymyxin B in patients with ESS, alongside an acceptable safety profile.
IMPLICATIONS: These results mark a significant milestone for the management of patients with septic shock, particularly regarding the use of adjunctive therapies for specific populations.
The TIGRIS trial was designed in collaboration with the FDA as part of the regulatory pathway to introduce the therapy into clinical practice in the United States for the management of septic shock.